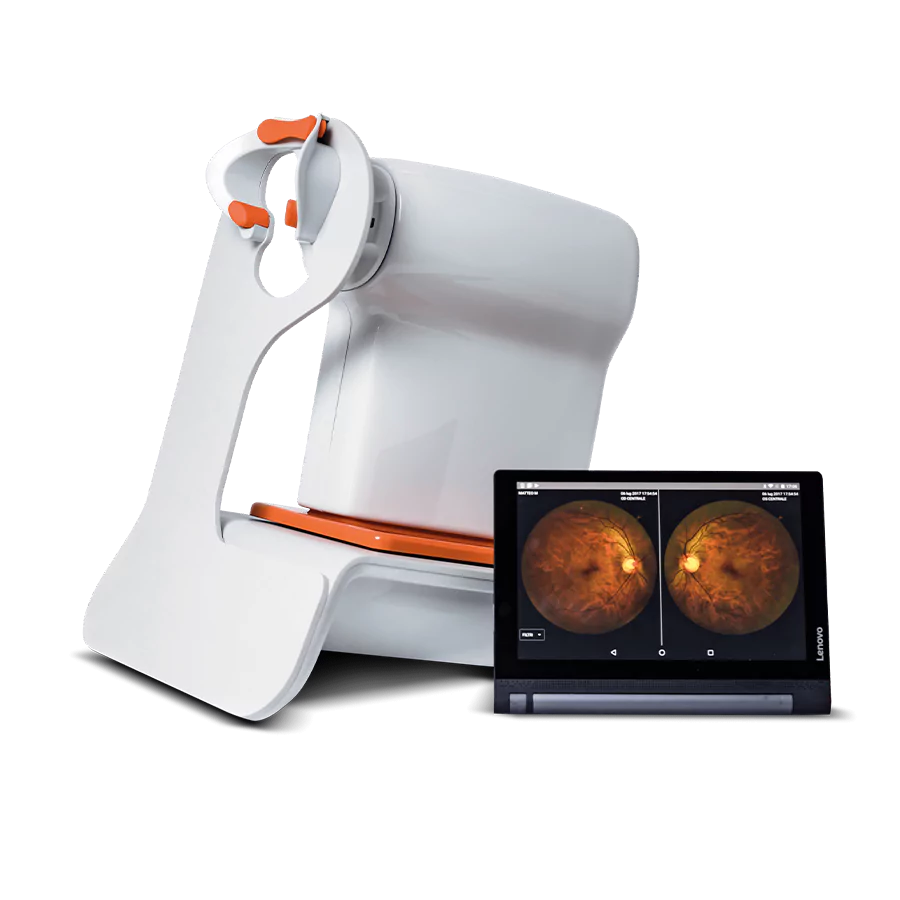
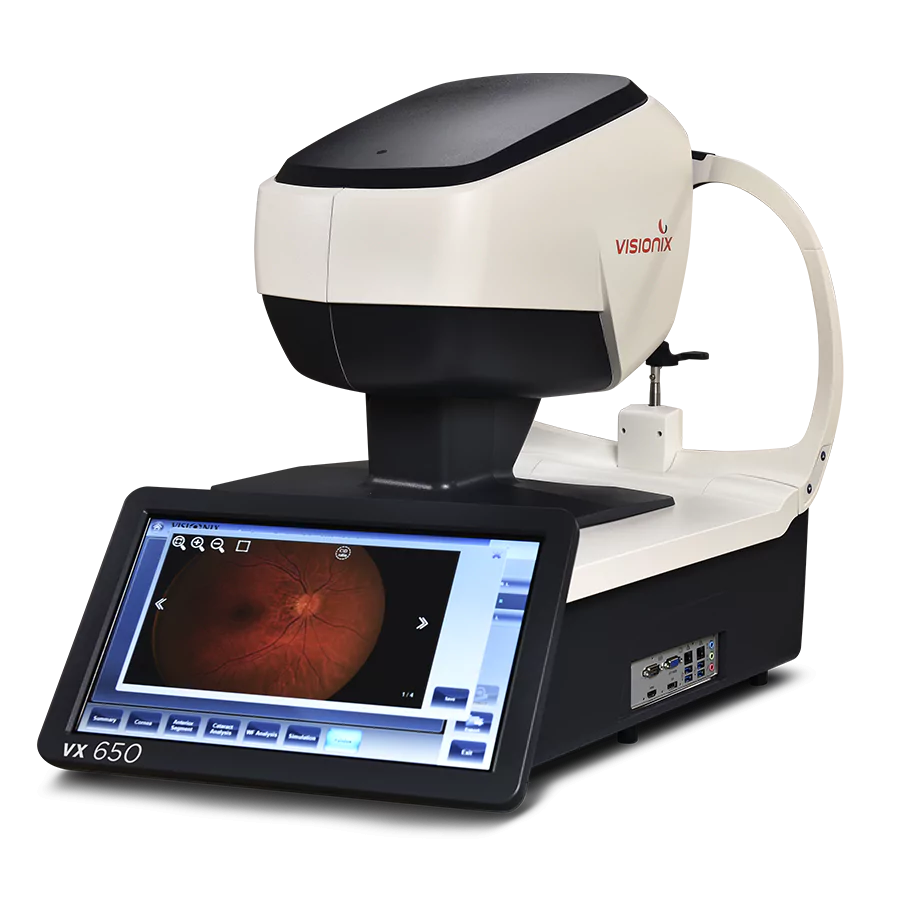
We create intelligent diagnostic, screening, refraction, and finishing lab equipment for Eye Care Professionals

Empower your practice with our intuitive, reliable, multimodal solutions for eye care professionals.
We’re a one-stop-shop that sells optometry systems that fit all needs and any budget. This means you can confidently make glasses on-site in your lab that are 100% accurate and error-free and will fit any patient comfortably.
With automated refraction, wellness OCTs, and automated screening equipment, your optometry practice can offer a blend of wellness and medical checkups to improve the care you offer to your patients and increase your revenue.
Streamline your ophthalmology practice and enhance patient consultations with our cutting-edge equipment. Experience a hassle-free workflow as our advanced technology empowers you to delegate data collection tasks to your skilled technicians.